货号:191-18375 产品名称:Stemsure® Serum Replacement(Stemsure®血清替代品)
可以用于ES细胞·iPS细胞培养用的血清代替品。血清含有分化诱导因子,会导致干细胞分化。使用本产品代替血清,可在ES和iPS细胞培养的同时,保持他们的未分化状态,达到稳定培养。同时本产品不含有剧毒物质。,将其作为普通物质处理。
使用本产品配制培养基培养的小鼠ES细胞,可用于制备嵌合体小鼠,并确认其种系传递。
制造商:FUJIFILM Wako Pure Chemical Corporation(富士胶片和光)
储存条件:-20°C
| 品牌 | 货号 | 产品名称 | 规格 |
| Wako | 191-18375 | Stemsure® Serum Replacement Stemsure®血清替代品 | 500 ml |
使用本产品配制培养基培养的小鼠ES细胞,可用于制备嵌合体小鼠,并确认其种系传递。
·特点 优势
·可用于嵌合体小鼠制备,并确认其种系传递。
·作为血清替代品,用于小鼠ES细胞·人IPS细胞培养。
·不含剧毒物质。
·案例 应用
1、嵌合体小鼠的制备与种系传递Update!!
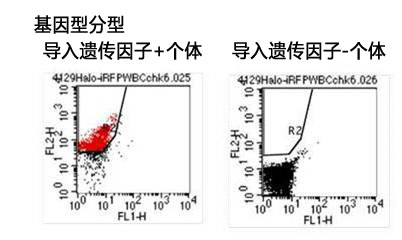
将组装HaloTag® cDNA的目标载体导入小鼠ES细胞,用含有Stemsure® Serum Replacement(SSR)的培养基培养。确认载体导入小鼠ES细胞后,将其注入到CR小鼠的胚盘胞,并移植至ICR小鼠的子言。得到的嵌合体小鼠与C57BL/6J小鼠交配,获得F1小鼠。
(1)嵌合体小鼠
收集嵌合体小鼠的血液,用TRITC(四甲基若丹明异硫氰酸盐)对HaloTag® 蛋白质染色。发现小鼠ES细胞源的HaloTag 的血液被染色,可确认为嵌合化。

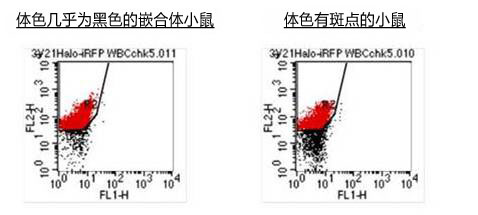
(2)F1小鼠
F1小鼠的血液发现HaloTag® 存在,可确认用SSR培养基培养的ES细胞嵌合小鼠能够正常存活。

2、人iPS细胞201B7的培养
(1)克隆形态与ALP染色
已确认可用StemSure® Serum Replacement(SSR)培养人iPS细胞201B7株。ALP染色呈阳性。

<培养基组成>
D-MEM/Ham's F-12+20%SSR +2 mmol/l L-Glutamine +1×MEM Non-essential Amino Acids +0.1
mmol/l StemSure® 2-Mercaptoethanol +1xPenicillin-Streptomycin +5 ng/ml bFGF
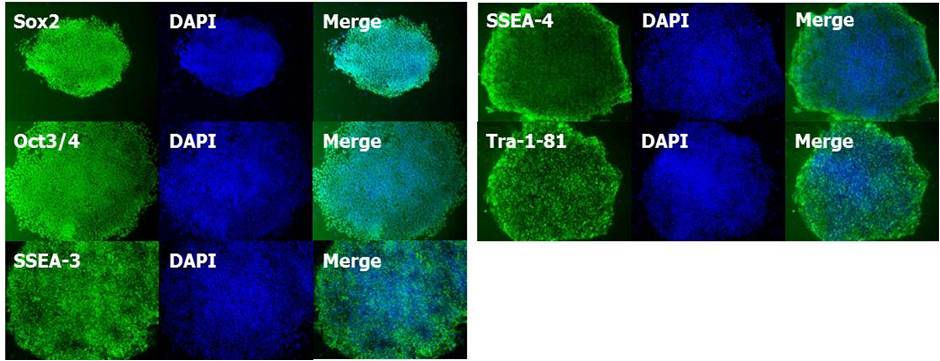
(2)未分化标记的发现
使用StemSure® Serum Replacement(SSR)培养的人iPS细胞201B7株,确认各种未分化标记(Sox2,Oct3/4,SSEA-3,SSEA-4,Tra-1-81)呈阳性。
3、小鼠ES细胞D3的培养
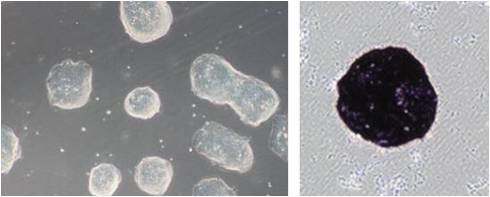
(1)细胞形态与ALP染色
使用StemSure® Serum Replacement(SSR)培养小鼠ES细胞D3,能观察到小鼠ES细胞群特有的光泽,ALP染色呈阳性。
(2)细胞增殖曲线
用不同培养基培养小鼠ES细胞D3进行14继代培养,比较细胞增殖。
如下图所示StemSure®系列与A公司有同等的性能。
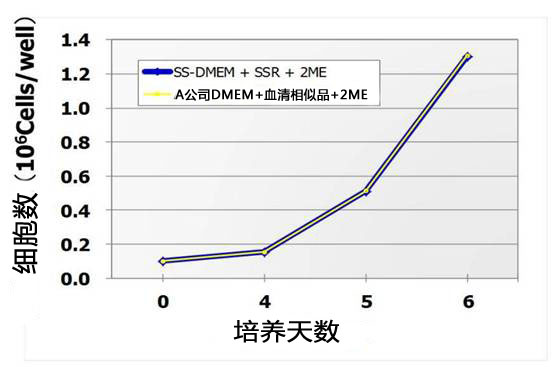
<StemSure®系列培养基组成>
StemSure®D-MEM+15%SSR +2 mmol/l L-Glutamine +1xMEM Non-essentialAmino Acids +0.1
mmol/lStemSure® 2-Mercaptoethanol+1×Penicillin-Streptomycin +1,000 units/ml StemSure®LIF
(使用胶原包被的12孔板)
(3)细胞群倍加数*
使用不同培养基对小鼠ES细胞D3进行继代培养,检查培养天数与细胞群倍加数的关系。
下图显示StemSure®系列与A公司产品的细胞群倍加数相同。
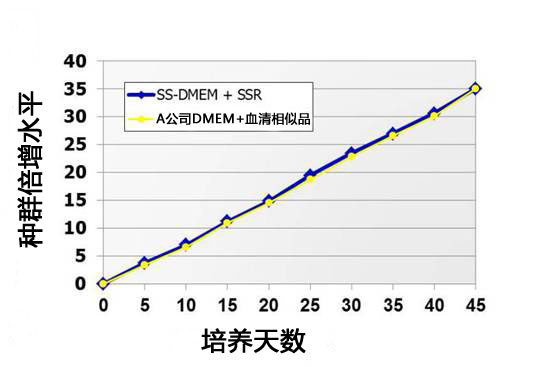
*细胞群倍加数cell population doubling level,缩写PDL,指从培养开始至现在细胞群的倍增次数。
<培养基组成>
StemSure® D-MEM+15%SSR+2 mmol/l L-Glutamine +1xMEM Non-essentialAmino Acids +0.1
mmol/l StemSure® 2-Mercaptoethanol +1×Penicillin-Streptomycin +1,000 units/ml StemSure® LIF
(使用胶原包被的12孔板)
(4)未分化标记的检测
使用StemSure®系列对小鼠ES细胞D3进行5代继代培养,各种未分化标记(Nanog,Oct3/4,Sox2,SSEA-1)呈阳性。
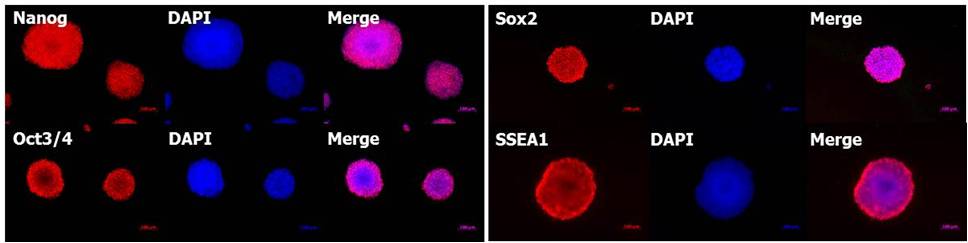
<培养基组成>
StemSure® D-MEM+15%SSR+2 mmol/l L-Glutamine +1×MEM Non-essential Amino Acids +0.1
mmol/l StemSure® 2-Mercaptoethanol+1xPenicillin-Streptomycin +1,000 units/ml StemSure® LIF
4、畸形瘤的形成
用StemSure®系列对小鼠ES细胞D3进行继代培养。对免疫不全小鼠的皮下注射培养后细胞。在形成皮下畸形瘤,其内部检测出神经组织(外胚叶源)、软骨组织(中胚叶源)、管腔结构纤毛上皮(内胚叶源)。
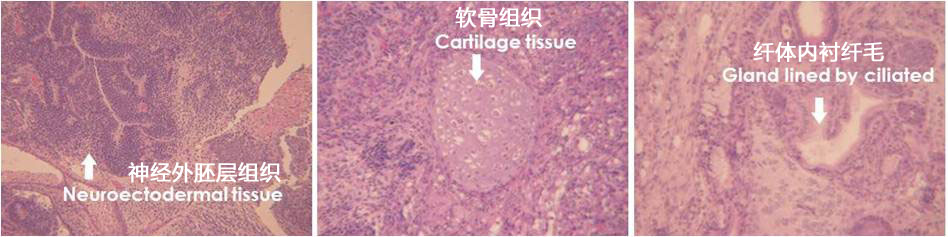
<培养基组成>
StemSure® D-MEM+15%SSR+2 mmol/l L-Glutamine +1×MEM Non-essential Amino Acids +0.1
mmol/l StemSure® 2-Mercaptoethanol+1xPenicillin-Streptomycin +1,000 units/ml StemSure® LIF
·参考文献
1. Obora,K, Onodera,Y, Takehara,T, Frampton,J, Hasei,J, Ozaki,T...& Fukuda,K.(2017).
Inflammation-induced miRNA-155 inhibits self-renewal of neural stem cells via suppression of CCAAT/enh ancer binding protein B(C/EBPB) expression. Scientific reports,7,43604.[ mouse neural stem cell (NS)]
2. Aihara,A, Koike,T, Abe,N, Nakamura,S, Sawaguchi,A, Nakamura,T.,..& Eto,K.(2017). Novel TPO receptor agonist TA-316 contributes to platelet biogenesis from human iPS cells. Blood acvances,17),
468-476.
3. Katayama,M, Kiyono,T, Kuroda,K, Ueda,K, Onuma,M, Shirakawa,H,& Fukuda,T.(2019). Rat-
derived feeder cells immortalized by expression of mutant CDK4, cyclin D, and telomerase can support stem cell growth. Biochimica et Biophysica Acta (88A)-Molecular Cel/Research,1866(5),945-956.
4. Katayama,M, Hirayama,T, Horie,K, Kiyono,T, Donai,K, Takeda,S.,..& Fukuda,T.(2016). Induced pluripotent stem cells with six reprogramming factors from prairie vole, which is an animal model for social behaviors. Ce/transplantation,25(5),783-796.[ prairie vole-derived iPsCs(pv-iPSCs)]
5.Kawata,M,Taniguchi,Y,Mori,D,Yano,F,Ohba,S,Chung,U.l,..&Saito,T.(2017).Different regulation of limb development by p63 transcript variants.PioS one,12(3),e0174122.【基因编辑小鼠iPS细胞】
6.Bui,P.L,Nishimura,K,Mondejar,G.S,Kumar,A,Aizawa,S,Murano,K,..&Ito,Y.(2019).Template Activating Factor-I a Regulates Retroviral Silencing during Reprogramming.Celf reports,29(7),1909-
1922.【Mouse embryonic fibroblasts(MEFs)】
7.Sumi,S,Kawagoe,M,Abe,R,Yanai,G,Yang,K.C,&Shirouzu,Y.(2017).A multiple-funnels cell culture insert for the scale-up production of uniform cell spheroids.Regenerative therapy,7,52-60.
8. Onozato,D, Yamashita,M, Nakanishi,A, Akagawa,T, Kida,Y, Ogawa,l,..& Matsunaga,T.(2018).
Generation of intestinal organoids suitable for pharmacokinetic studies from human induced pluripotent stem cells. Drug Metabolism and Disposition,46(11),1572-1580.
9. Hayashi,Y, Matsumoto,J, Kumagai,S, Morishita,K, Xiang,L, Kobori,Y,..& Sumaru,K.(2018).
Automated adherent cell elimination by a high-speed laser mediated by a light-responsive polymer.
Communications biology,1(1),218.[ hiPsc differentiation]
10. Osafune,K, Toyoda,T, Takekawa,S, Nakamura,G,& Ito,R.(2019).U.S. Patent Application No.6/490,394.
11. Kato,T, Kanemura,Y, Shofuda,T,& Fukusumi,H.(2016).U.S. Patent Application No.74/900,975.
12. Eitoku,M, Kato,H, Suganuma,N,& Kiyosawa,H.(2018). Markers associated with neuron-specific Ube3a imprinting during neuronal differentiation of mouse embryonic stem cells. Cytotechnology,70(1),45-53.
13. Onozato,D, Yamashita,M, Fukuyama,R, Akagawa,T, Kida,Y, Koeda,A...& Matsunaga, T.(2018).
Efficient generation of Cynomolgus monkey induced pluripotent stem cell-derived intestinal organoids with pharmacokinetic functions. Stem celis and devefopment,27(15),1033-1045.
14. Katayama,M, Hirayama,T, Kiyono,T, Onuma,M, Tani,T, Takeda,S...& Fukuda,T.(2017).
Immortalized prairie vole-derived fibroblasts (VMF-K4DTs) can be transformed into pluripotent stem cells and provide a useful tool with which to determine optimal reprogramming conditions. Journat of Reproduction and Development,2016-164.
15. Matsuyama,A,& Okura,H.(2017).U.S. Patent No.9,644,181. Washington, DC:U.S. Patent and Trademark Office.
16. Tomizawa,M.(2016). Culture Medium And Method For Inducing Differentiation of Pluripotent Stem Cells To Hepatoblasts.U.S. Patent Application No.14/749,715.